Collection, Transport & Preservation of Clinical Specimens Containing Viruses
UTM® Universal Transport Medium™ is the leading transport medium for stabilization and transport of unprocessed upper respiratory specimens containing respiratory virus nucleic acids. It is designed for use with compatible molecular assays.
Copan Universal Transport Medium (UTM®) System is intended for the stabilization and transportation of an unprocessed upper respiratory clinical specimen suspected of containing:
Downstream Applications:
*Consult with the assay manufacturer’s instructions for use for compatibility and specific target analyte processing directions.
Copan has received FDA clearance (510(k) number K232357), which updates the intended use of UTM® Universal Transport Medium™ without beads for molecular testing applications.

UTM® is room temperature stable, eliminating the need for refrigeration storage prior to collection.

UTM® Universal Transport Medium™ preserves the viability of all target organisms tested for 48 hours at both room and refrigerated temperatures; it can also be stored at -80°C for one year.
A phenol red pH indicator provides a visual gauge of UTM® Universal Transport Medium™ integrity throughout the storage and life of the product.

Copan UTM® has a unique formulation which includes proteins for virus stabilization, antibiotics and antimycotics that inhibit bacterial and fungal flora, keeping your sample free of unwanted growth without affecting target organisms.
Clinical studies demonstrate that UTM® Universal Transport Medium™, when paired with FLOQSwabs®, is equivalent to collections using aspirates and nasal washes for the diagnosis of respiratory virus infection. UTM® Universal Transport Medium™ paired with Copan FLOQSwabs® provide a cost-effective alternative to traditional invasive methods, improving patient comfort. [1]
UTM® delivers 48 hours of room temperature stability without refrigeration, maintaining organism viability while complying with CLSI M40-A2 guidelines. The formulation contains includes proteins for virus stabilization, antibiotics and antimycotics to inhibit bacterial and fungal overgrowth, securing pathogens of interest. A phenol red pH indicator visually confirms media integrity over extended transport and storage.
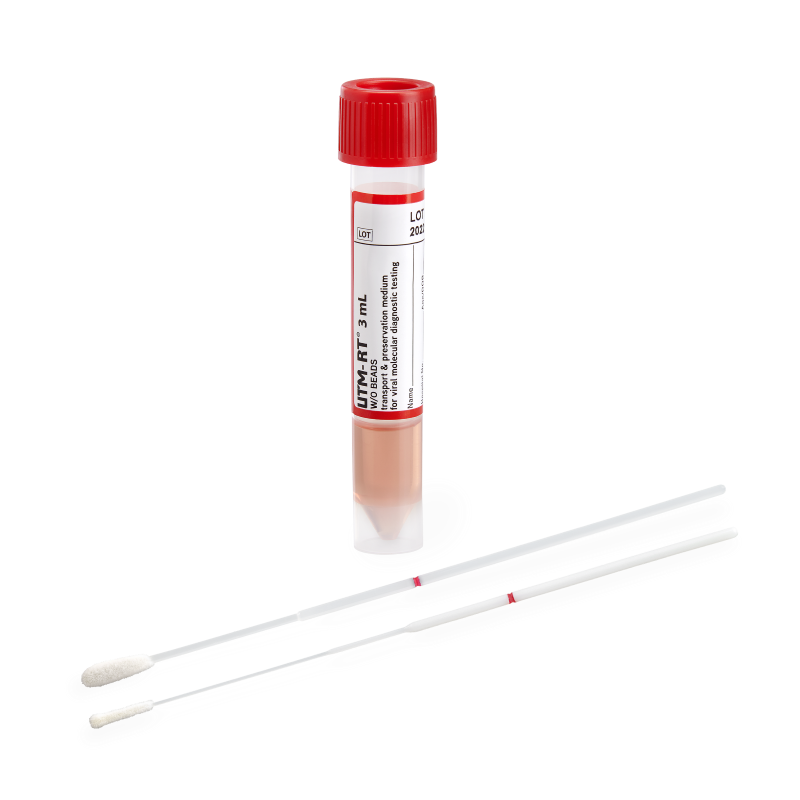
Choose from bulk media tubes or kits paired with a FLOQSwabs® flocked swab. Self-standing shatterproof polypropylene conical tubes ensure ease of use for clinicians, laboratory staff, and automated platforms.
UTM® is compatible with downstream antigen detection and molecular-based assays and can be processed manually or with automated laboratory platforms such as Copan UniVerse®.

A new format of Copan's well-known UTM® is now available. The new product configuration allows for direct loading on Roche cobas® 5800/6800/8800 systems, supporting the fully validated workflow. In addition, this new format includes a formal claim for its use for the molecular detection of upper respiratory pathogens.

UTM® Universal Transport Medium™ is the leading transport medium for stabilization and transport of unprocessed upper respiratory specimens containing respiratory virus nucleic acids. It is designed for use with compatible molecular assays.
Copan Universal Transport Medium (UTM®) System is intended for the stabilization and transportation of an unprocessed upper respiratory clinical specimen suspected of containing:
Downstream Applications:
*Consult with the assay manufacturer’s instructions for use for compatibility and specific target analyte processing directions.
There are no results matching your query.
3 mL UTM® Universal Transport Medium™
Tube: 16x100mm
Cap: Red Cap
Bulk Packaged
50/Pack; 6 Packs/Case
3 mL UTM® Universal Transport Medium™
Swab: Nylon® Flocked Swab
Tip: Flexible Minitip
Breakpoint: 100mm
Tube: 16x100mm
Cap: Red Cap
Individually Packaged,
Sterile 50/Pack; 6 Packs/Case
3 mL UTM® Universal Transport Medium™ w/o beads
Swab: Nylon®Flocked Swab
Tip: Regular
Breakpoint: 100mm
Tube: 16x100mm
Cap: Red Cap
Individually Packaged,
Sterile 50/Pack; 6 Packs/Case
3 mL UTM® Universal Transport Medium™ w/o beads
Swab: Nylon® Flocked Swab
Tip: Minitip
Breakpoint: 80mm
Tube: 12x80mm
Cap: Red Cap
Packaged in Zip Top Plastic Bag, 100 Swabs/Box
3 mL UTM® Universal Transport Medium™
Tube: 12x80mm
Cap: Red Cap
Bulk Packaged
50/Pack; 6 Packs/Case
3 mL UTM® Universal Transport Medium™
Tube: 15.5x95mm
Cap: Blue Cap
Bulk Packaged
50/Pack; 6 Packs/Case
Swab: Nylon® Flocked Swab
Tip: Minitip
Breakpoint: 80mm
Tube: 15.5x95mm
Cap: Blue Cap
Individually Packaged, Sterile
50/Box; 6 Boxes/Case
Swab: Nylon® Flocked Swab
Tip: Regular
Breakpoint: 80mm
Tube: 15.5x95mm
Cap: Blue Cap
Individually Packaged, Sterile
50/Box; 6 Boxes/Case
This video is intended for nurses, lab technicians and other clinical staff, and provides simple step-by-step instructions for using Copan's flocked swabs (FLOQSwabs®) and UTM® for nasopharyngeal sample collection.
This video is intended for nurses, lab technicians and other clinical staff, and provides simple step-by-step instructions for using Copan's flocked swabs FLOQSwabs® for eye specimen collection.
This video is intended for nurses, lab technicians and other clinical staff, and provides simple step-by-step instructions for using Copan's flocked swabs (FLOQSwabs®) and UTM® for nasal sample collection.
[2] Consult with the assay manufacturer’s instructions for use for compatibility and specific target analyte processing directions
Copan specializes in working with partners in assay manufacturing, microbiome research, and at home collection kit developers and more to create customized swabs and kits, including filling of tubes with custom reagents. Contact us today to speak to a knowledgeable Copan team member.