Collection, Transport & Preservation for Aerobic, Anaerobic and Fastidious Bacteria
Streamline clinical sampling with Copan's patented ESwab® kits, powered with innovative FLOQSwabs® technology. ESwab® combines a Copan-invented flocked swab with 1mL of Liquid Amies in a plastic, screw cap tube.
The innovative system elutes over 90% of patient specimen into the liquid medium1. The result is evidence-based, improved pathogen recovery, expanded testing capabilities, and better patient care.2, 3, 4, 11
ESwab® Collection, Transport & Preservation System for Aerobic, Anaerobic and Fastidious Bacteria:6, 7, 9
Multiple investigations can be performed from the same ESwab® sample including:2, 3, 7, 8, 10
ESwab® has been tested and validated in full compliance with CLSI M40-A2: Quality Control of Microbiological Transport System Standard.
ESwab® kits contain FLOQSwabs® which have no internal core to trap the sample, allowing for rapid sample uptake and instantaneous elution of over 90% of the sample into the liquid medium.1, 4, 5

ESwab® is a collection and transport system FDA cleared for aerobic, anaerobic and fastidious bacteria, maintaining viability for up to 48 hours at room or refrigerator temperature 6

ESwab® samples are easily processed on automated specimen processors, and automatic pipettors minimizing manual handling and maximizing investment in automation.2

ESwab® is validated for molecular and rapid antigen testing on an increasing number of manufacturers' platforms.7, 8
ESwab® has been tested and validated in full compliance with CLSI M40-A2: Quality Control of Microbiological Transport System Standard.
The FDA-cleared collection and transport system is suitable for aerobic, anaerobic, and fastidious bacteria maintaining viability for up to 48 hours at room or refrigerator temperature (Neisseria gonorrhoeae survival at 24 hours per CLSI standard).6
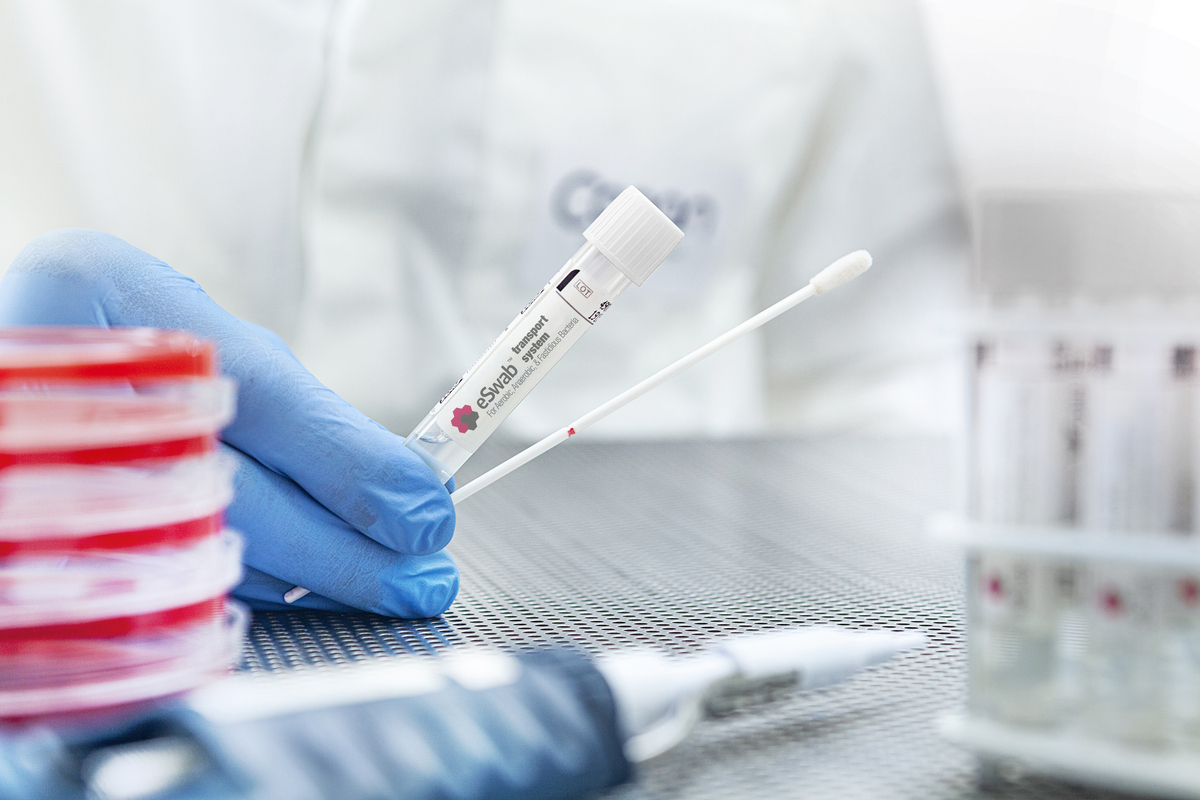
For laboratories, ESwab® provides a broad range of testing applications, replacing the traditional practices requiring multiple swabs with just one ESwab® sample, thus eliminating costs associated with stocking numerous swab types.1, 3, 13
For patients, this means fewer samples need to be collected, offering a more comfortable experience.2
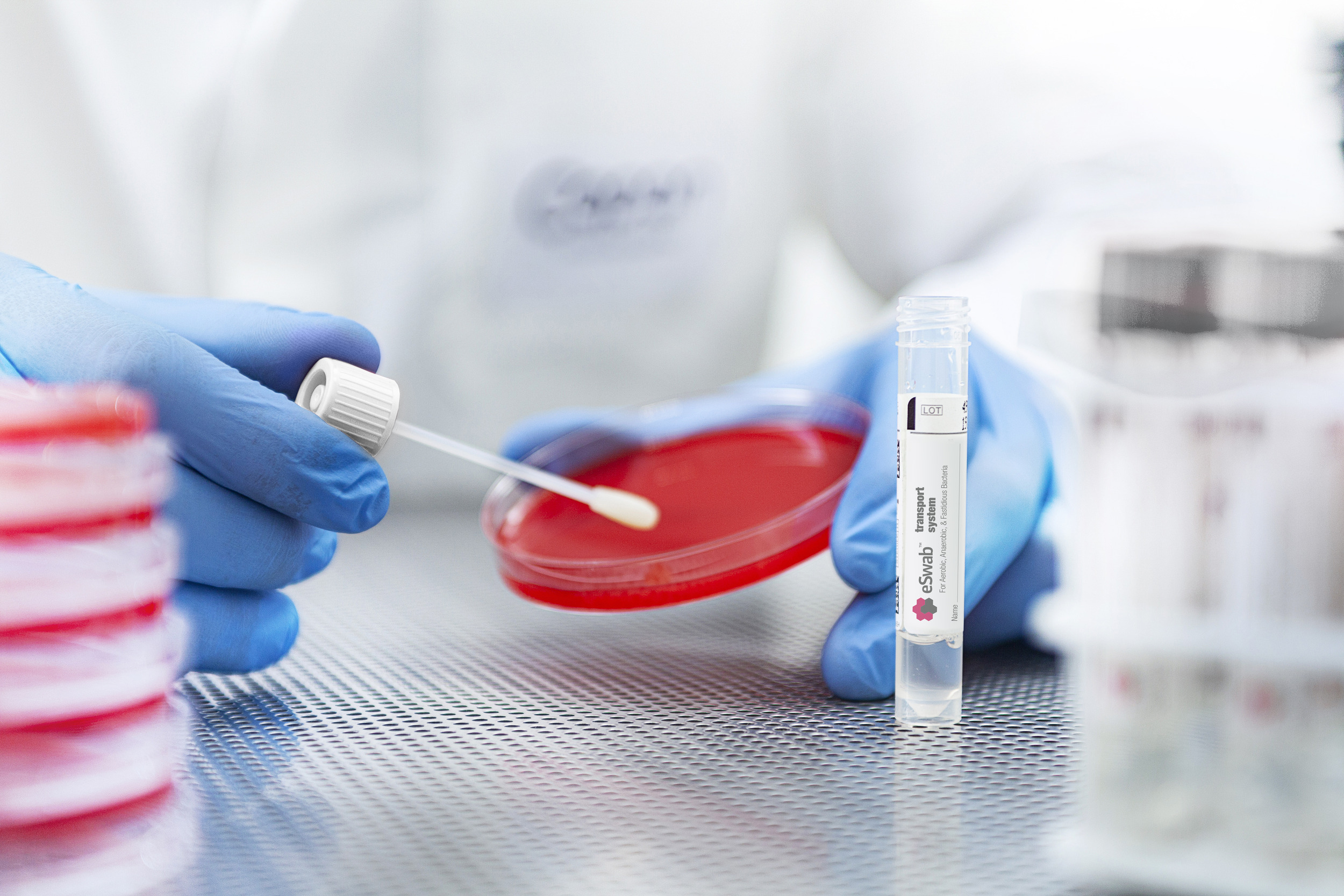
ESwab® provides aliquots for culture plates, molecular assays, rapid antigen tests, gram stains, and more - revolutionizing efficiency with extended storage and seamless processing.
Experience improved pathogen recovery, expanded testing capabilities, and better patient care in a single, versatile kit.1

The anatomically designed FLOQSwabs® within the ESwab® kit balance patient comfort while providing optimized sample collection and transport for improved assay sensitivity and expanded testing capabilities.2, 3, 4, 5
Paired with ESwab® transport media, FLOQSwabs® enable complete pathogen release and consistent, measurable volume for automated processors - easing collection, improving recovery and sensitivity across bacteriology and virology assays. 2

Streamline clinical sampling with Copan's patented ESwab® kits, powered with innovative FLOQSwabs® technology. ESwab® combines a Copan-invented flocked swab with 1mL of Liquid Amies in a plastic, screw cap tube.
The innovative system elutes over 90% of patient specimen into the liquid medium1. The result is evidence-based, improved pathogen recovery, expanded testing capabilities, and better patient care.2, 3, 4, 11
ESwab® Collection, Transport & Preservation System for Aerobic, Anaerobic and Fastidious Bacteria:6, 7, 9
Multiple investigations can be performed from the same ESwab® sample including:2, 3, 7, 8, 10
There are no results matching your query.
1 mL Liquid Amies Medium
Swab: Nylon® Flocked Swab
Tip: Regular
Breakpoint: 80mm
Tube: 12x80mm
Cap: White Capture Cap
Individually Packaged, Sterile
50/Pack; 10 Packs/Case
1 mL Liquid Amies Medium
Swab: Nylon® Flocked Swab
Tip: Minitip
Breakpoint: 80mm
Tube: 12x80mm
Cap: Green Capture
Individually Packaged, Sterile
50/Pack; 10 Packs/Case
1 mL Liquid Amies Medium
Swab: Nylon® Flocked Swab
Tip: Flexible Minitip
Breakpoint: 100mm
Tube: 12x80mm
Cap: Blue Non-Capture Cap
Individually Packaged,
Sterile 50/Pack; 10 Packs/Case
Double Swab Kit;
1 mL Liquid Amies Medium; Dry Tube
Swabs: Nylon® Flocked Swab; Rayon Swab
Tips: Regular
Breakpoints: 80mm; No Breakpoint
Tubes: 12x80mm Tube w/ media.; Round Bottom w/o media
Individually Packaged, Sterile
50/Pack; 10 Packs/Case
1 mL Liquid Amies Medium
Swab: Nylon® Flocked Swab
Tip: Regular
Breakpoint: 80mm
Tube: 12x80mm
Cap: Purple Non-Capture
Individually Packaged, Sterile
50/Pack; 10 Packs/Case
Double Swab Kit;
1 mL Liquid Amies Medium
Swabs: 2 Nylon® Flocked Swabs
Tips: Regular
Breakpoints: 80mm; No Breakpoint
Cap: White Capture
Tube: 12x80mm Tube
Individually Packaged, Sterile
50/Pack; 8 Packs/Case
Triple Swab Kit;
1 mL Liquid Amies Medium
Swabs: Nylon® Flocked Swabs
Tips: Regular
Breakpoints: One 80mm; Two No Breakpoint
Tubes: 12x80mm Tube w/ media; Round Bottom w/o media
Cap: White Capture
Individually Packaged, Sterile
50/Pack; 8 Packs/Case
This video is intended for nurses, lab technicians, and other clinical staff, and provides simple step-by-step instructions for using Copan’s ESwab® for sample collection.
This video demonstrates how to perform planting and streaking using Copan's ESwab® (Pipet planting and Streaking)
Copan's new ESwab® plastic applicator material and new breaking technique video is a visual guide to demonstrate how to recognize the new material, and how to perform the new breaking technique.
1. Copan Group. Internal elution studies, n.d.
2. Fontana C, et al. Adv Microbiol. 2013;3:504-510. How Liquid Based Microbiology Can Change the Workflow.
3. Bjerre RD, et al. Sci Rep. 2019;9:17287. Effects of sampling strategy and DNA extraction on human skin microbiome investigations.
4. Tan TY, et al. Pathology. 2014;46(3):230-233. Evaluation of bacterial recovery and viability.
5. Van Horn KG, et al. Diagn Microbiol Infect Dis. 2008;62(4):471-475. Comparison of 3 swab transport systems.
6. Copan Italia S.p.A. ESwab® Collection and Transport System. 2023.
7. Van TT, et al. J Clin Microbiol. 2017;55(4):1147-1153. Prevalence of Fusobacterium necrophorum in Children Presenting with Pharyngitis.
8. Beck E, et al. Open Forum Infect Dis. 2018;5(Suppl 1). Evaluation of Three Rapid Molecular Assays.
9. Tyrrell KL, et al. J Clin Microbiol. 2016;54(5):1364-1367. Comparison of the Copan eSwab System with an Agar Swab Transport System.
10. Fontana C, et al. BMC Res Notes. 2009;2:244. Comparison of the eSwab collection and transportation system to an amies gel transystem.
11. Yarbrough ML, et al. J Clin Microbiol. 2018;56(1). Multicenter Evaluation of the Xpert MRSA NxG Assay.
Copan specializes in working with partners in assay manufacturing, microbiome research, and at home collection kit developers and more to create customized swabs and kits, including filling of tubes with custom reagents. Contact us today to speak to a knowledgeable Copan team member.