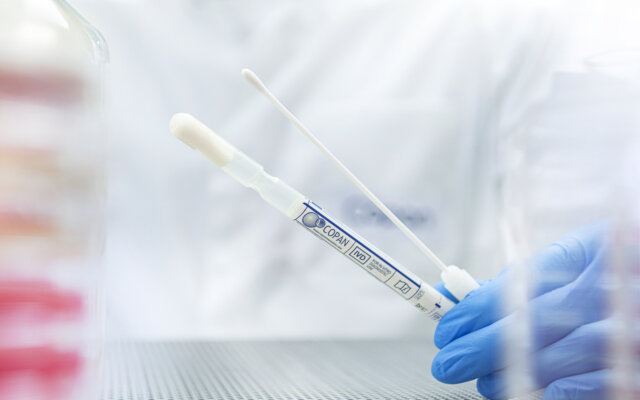
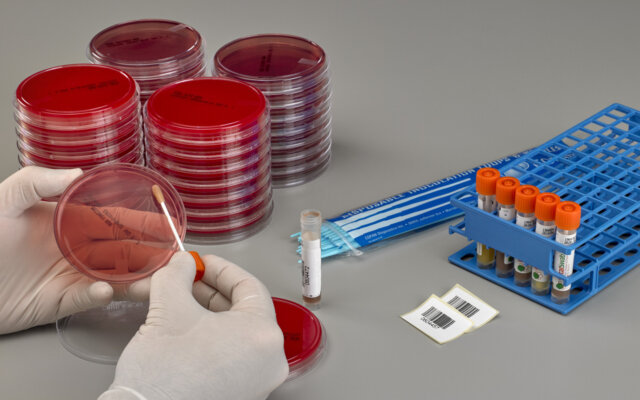
Sample Collection & Transport Kits
Copan's extensive range of collection and transport kits feature patented flocked or fiber wrapped swabs paired with specialized transport media to safely collect specimens and maintain sample integrity for downstream testing using culture-based and molecular methods.
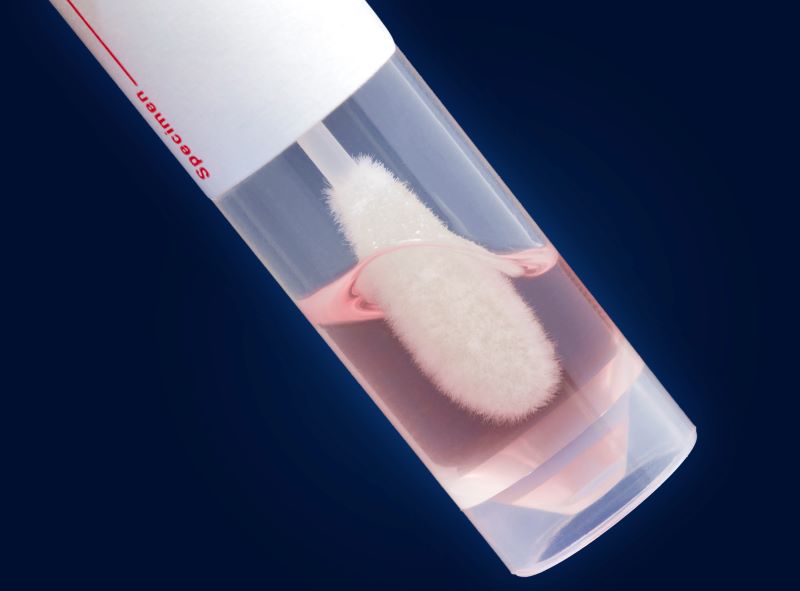
UTM® Universal Transport Medium™
Copan's UTM® Universal Transport Medium™ kits feature Copan’s patented FLOQSwabs® flocked swabs paired with specialized medium in tubes to reliably collect and safely transport clinical specimens for sensitive molecular and rapid antigen detection of viruses, Chlamydia, Mycoplasma and Ureaplasma.
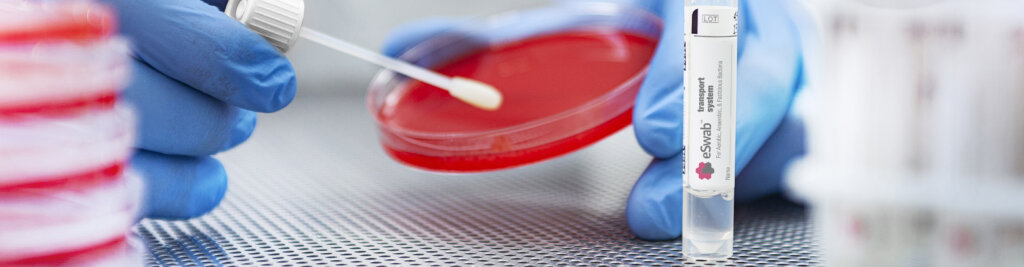
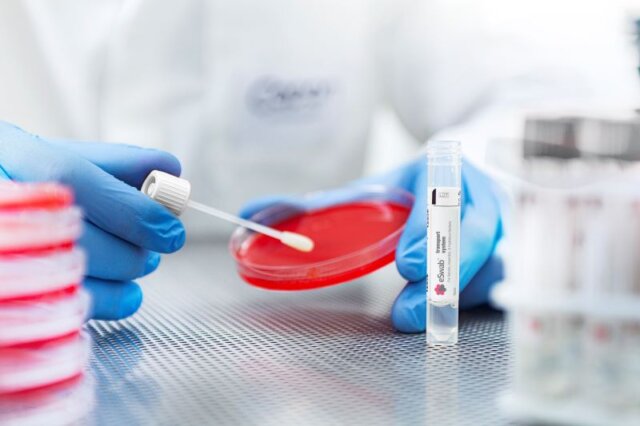
ESwab® Liquid Based Collection and Transport
ESwab® is Copan's innovative multipurpose specimen collection, and transport system pairing our patented FLOQSwabs® with Liquid Amies Medium for superior pathogen recovery and versatile utilization for microbiology assays for aerobic, anaerobic and fastidious bacteria.
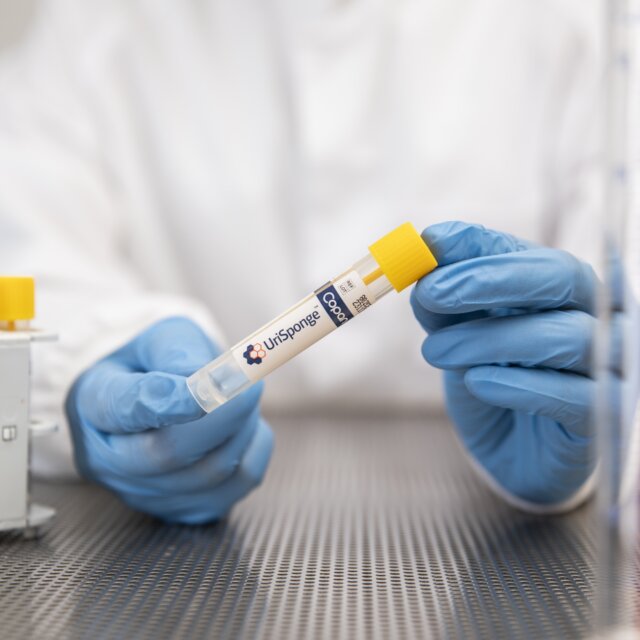
UriSponge® System for Urine Specimens
UriSponge® is Copan’s FDA-cleared, innovative device for urine specimen collection, transport, and preservation, designed to enhance workflow efficiency, reduce safety risks, and maintain specimen integrity.
FecalSwab® System for Enteric Bacteria
FecalSwab® simplifies stool collection by pairing our FLOQSwabs® flocked swabs with Cary-Blair medium to liquify samples for improved recovery of enteric bacteria and viruses.
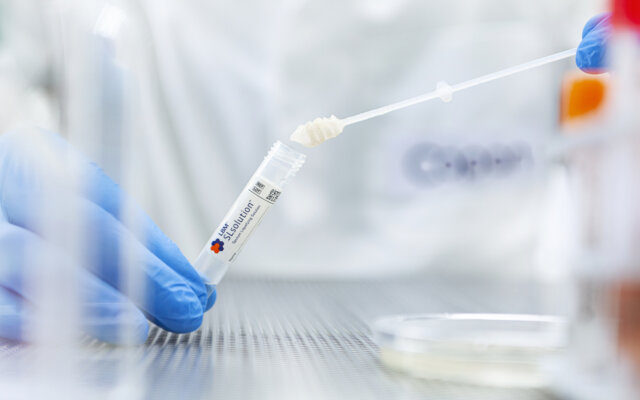
SnotBuster™ Sputum Liquefying System
The SnotBuster™ liquefies sputum samples prior to culturing for the isolation of bacteria and fungi, without affecting morphology, growth or microscopic staining. Easily transfer sputum samples using Copan invented Sputum Dipper™, a unique drill-shaped tool for managing challenging sputum samples.
eNAT® System for Molecular Specimens
eNAT® pairs Copan's patented FLOQSwabs® with a guanidine medium that inactivates pathogens while stabilizing nucleic acids, enabling prolonged room temperature storage of viruses, bacteria, chlamydia, trichomonas vaginalis, and mycoplasma prior to extraction and analysis molecular assays.
MSwab® System for Molecular Assays
MSwab® medium provides homogeneous aliquots of sample suspension to run multiple tests from the same tube. In the laboratory, specimens collected in MSwab® can be analyzed using standard clinical procedures for bacterial culture of aerobic and facultative anaerobic gram-positive microorganisms, Viral culture of HSV 1 HSV 2 viruses, and viral and bacterial nucleic acid detection.

Transystem™ Traditional Bacteriology Transport
With an extensive range of traditional bacteriology, fiber wrapped swab systems, the Transystem™ product line comprises of Amies, Stuart, and Cary-Blair media in agar gel and liquid formats to serve sample collection and transport needs in clinical bacteriology.