Copan FecalSwab® Brochure
See how FecalSwab®, Copan's patented sample collection and preservation system for enteric bacteria, fits into the broader Liquid Based Microbiology (LBM) family. Preview the brochure below or download the PDF to share with your laboratory.
What's inside
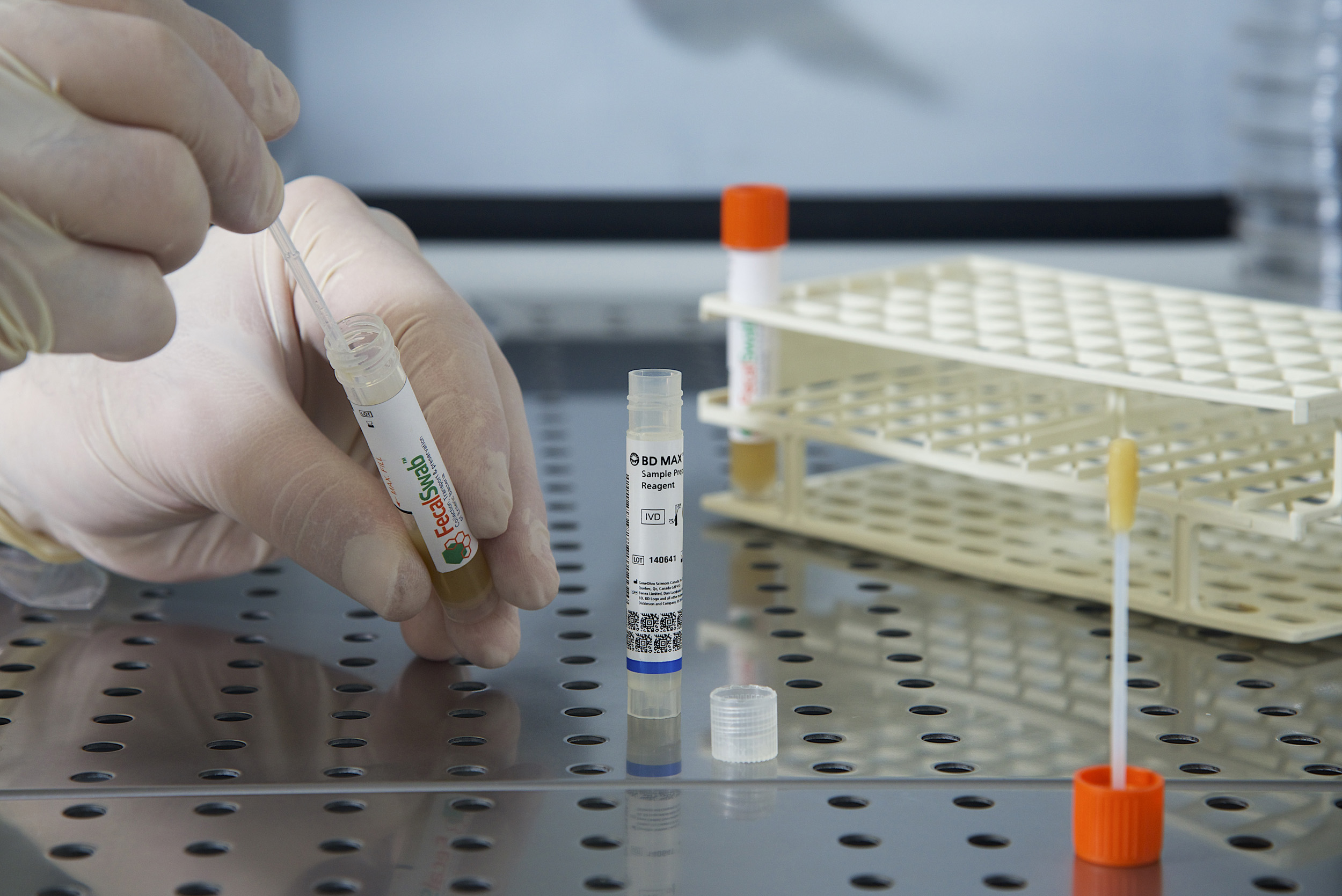
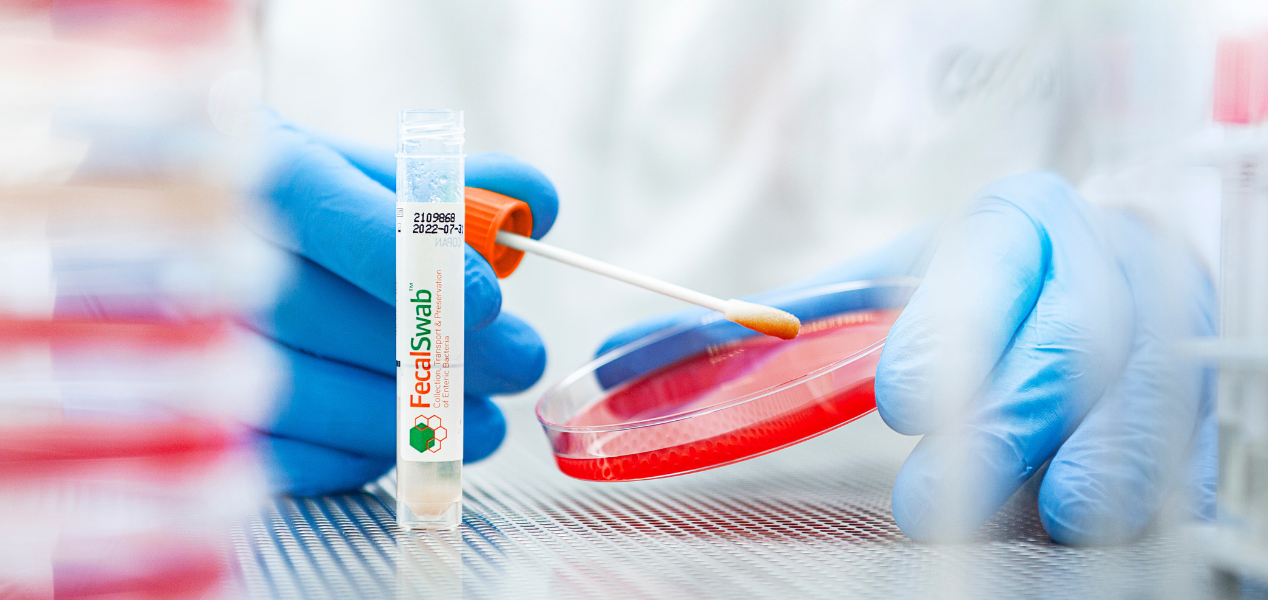
A look inside Copan’s Liquid Based Microbiology brochure, with a focus on FecalSwab®, the patented system that pairs FLOQSwabs® technology with 2 mL of Cary-Blair medium to convert challenging stool specimens into easy-to-process liquid specimens.
- •FDA cleared for enteric pathogen recovery: bacterial and viral recovery via traditional culture, with documented compatibility on enteric molecular assays where Cary-Blair transport is specified in the assay package insert
- •Molecular clearance: FDA cleared for use with the BD MAX™ Enteric Bacterial Panel and BD MAX™ Extended Enteric Bacterial Panel
- •Cleaner workflow than bulk containers: smaller specimen volume eliminates the need to vent the container and prevents messy accidents during processing, with compact tubes that ship more efficiently than traditional bulky fecal containers
- •Better pediatric collection: rectal swab option simplifies pediatric specimen collection versus collecting from diapers, where diaper material can interfere with some assays
- •Faster time to treatment: rectal swab collection means clinicians can collect a specimen immediately, without waiting for the patient to produce a fecal specimen
- •Standardized rectal collection: optional anatomically designed stopper ensures the swab tip reaches the transition zone of the rectum for consistent rectal swab specimen collection
- •Automation ready: liquid-format stool specimens are easily processed on automated specimen processors and pipettors, minimizing manual handling and the mess that comes with it
The full brochure also covers ESwab®, SnotBuster™, and UriSponge®, plus how Liquid Based Microbiology opens the door for automated specimen processing on WASP®.
Always read the manufacturer’s package insert for specific instructions regarding specimen collection and transport for the type of test kit being used. Specimen collection should be performed by health care personnel who have completed training and demonstrated competency. Product availability and regulatory approval may vary by country or region.
Need help?
Considering liquid-based stool collection and transport for your enteric workflow? Our team can walk you through assay compatibility, pediatric and rectal collection options, and what’s involved in transitioning from traditional bulk fecal containers to FecalSwab®.
Whether you’re evaluating FecalSwab® for a single workflow or rolling it out across your laboratory in support of automation on WASP®, we’re here to help with verification guidance, training, and change management.