Copan ESwab® Brochure
See how ESwab®, Copan's patented liquid-based collection and transport system, fits into the broader Liquid Based Microbiology (LBM) family. Preview the brochure below or download the PDF to share with your laboratory.

What's inside
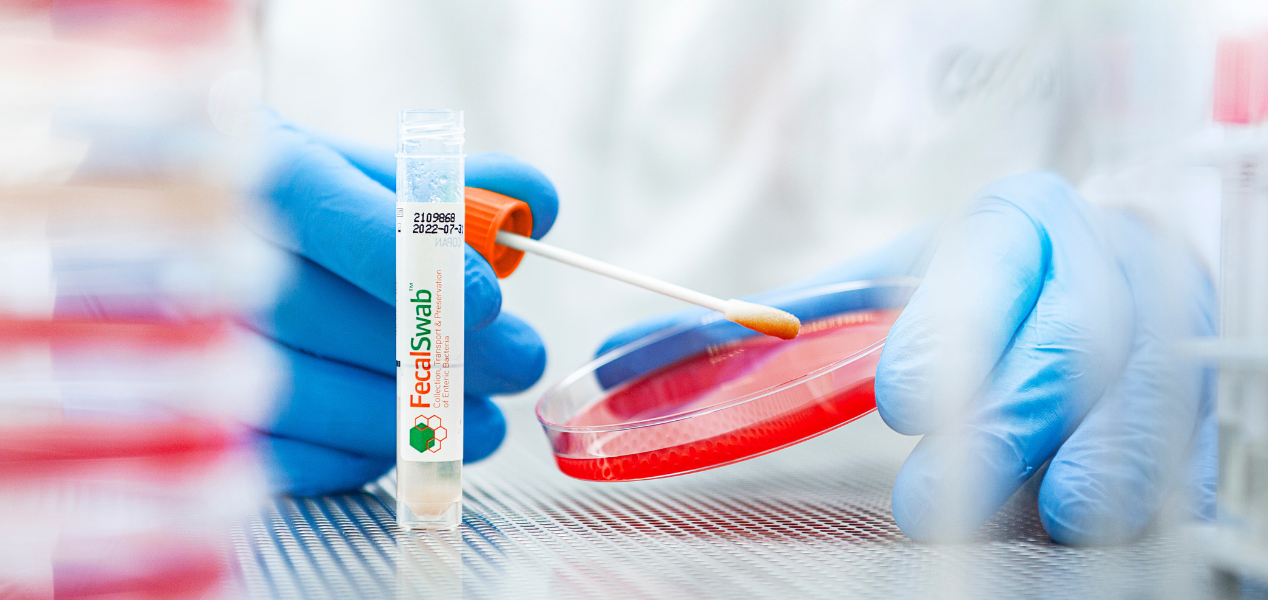
A look inside Copan’s Liquid Based Microbiology brochure, with a focus on ESwab®, the patented collection and transport system that pairs FLOQSwabs® technology with 1 mL of Liquid Amies medium for quantitative, multi-purpose specimen collection.
- •FDA cleared for aerobic, anaerobic, and fastidious bacteria: maintains organism viability for up to 48 hours at room or refrigerator temperature (N. gonorrhoeae survival 24 hours per CLSI standard)
- •More sample, more answers: elutes over 90% of the patient specimen into the Liquid Amies medium, supporting multiple investigations from a single collection (culture plates, Gram stains, rapid antigen tests, and molecular assays)
- •Better diagnostic signal: improved pathogen recovery for traditional bacteriology culture, with a homogeneous sample for more consistent and precise Gram stains
- •Validated across platforms: tested for molecular and rapid antigen assays on many manufacturers’ platforms; always confirm compatibility in the assay’s package insert
- •Automation ready: easily processed on automated specimen processors and liquid-handling pipettors, minimizing manual handling and maximizing return on automation investment
- •Three swab formats: regular, minitip, and flexible minitip applicators for nasal, throat, vaginal, rectal, wound, urogenital, pediatric, and nasopharyngeal collection
The full brochure also covers FecalSwab®, SnotBuster™, and UriSponge®, plus how Liquid Based Microbiology opens the door for automated specimen processing on WASP®.
Always read the manufacturer’s package insert for specific instructions regarding specimen collection and transport for the type of test kit being used. Specimen collection should be performed by health care personnel who have completed training and demonstrated competency. Product availability and regulatory approval may vary by country or region.
Need help?
Considering a switch to Liquid Based Microbiology (LBM)? Our team can walk you through swab format selection, assay compatibility, and what’s involved in transitioning from traditional fiber swabs to ESwab®.
Whether you’re evaluating ESwab® for a single workflow or rolling it out across your laboratory in support of automation on WASP®, we’re here to help with verification guidance, training, and change management.