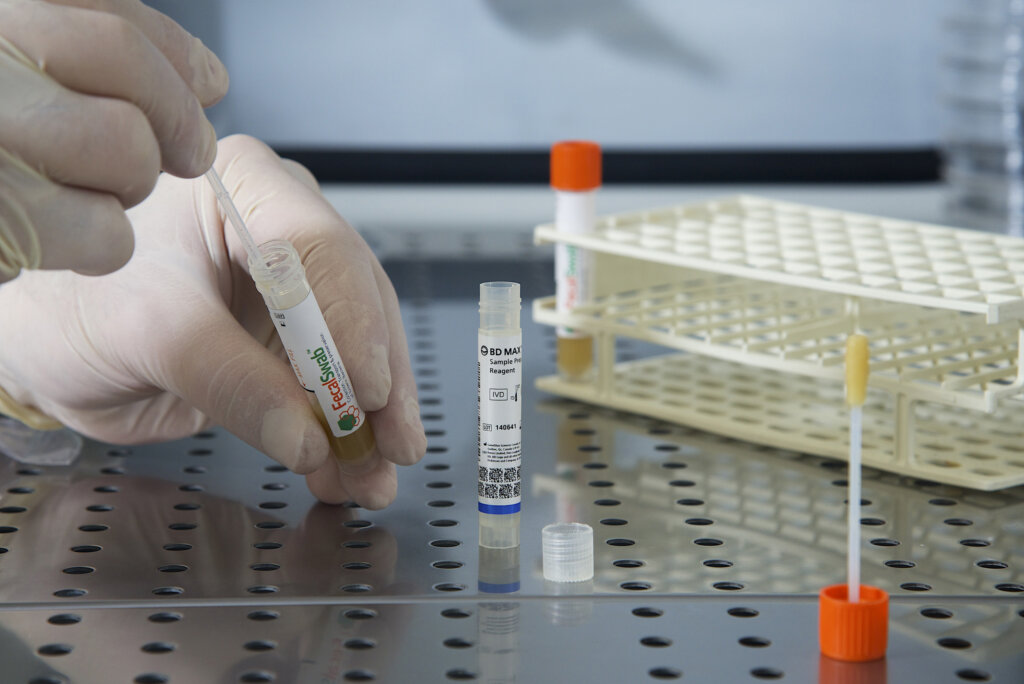
Study Validates Effectiveness of Copan FecalSwab® for Enteric Pathogen Detection on the BD MAX™ System
 Read More
Read More
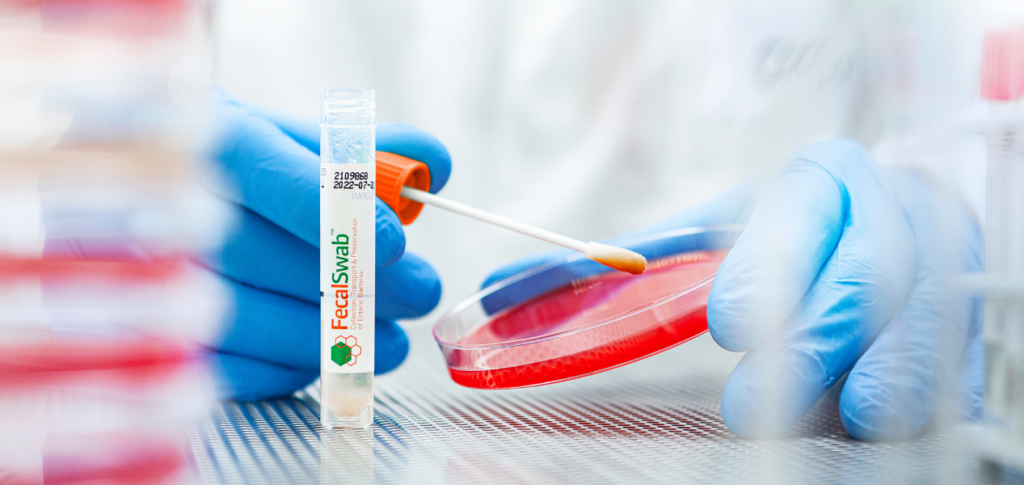
Comprehensive Guide to Cary-Blair Transport Medium: The Gold Standard for Fecal Sample Collection and Transport
 Read More
Read More
Study Presented at CACMID 2024 Highlights Copan's UniVerse® as a Game-Changer in Clinical Molecular Specimen Preparation
 Read More
Read More
Study Demonstrates Effectiveness of eNAT® Medium for Long-Term Fecal Microbiome Sample Preservation
 Read More
Read More
New Study Shows Copan's Colibrí™ Automates MALDI-TOF Sample Preparation with High Accuracy
 Read More
Read More
Research Roundup: Sample Collection for COVID: How Does Saliva Stack up Against Swabbing?
 Read More
Read More
Copan Expands U.S. Manufacturing Footprint with Multimillion-Dollar Investment
 Read More
Read More
Copan Diagnostics Announces Leadership Transition in Medical and Scientific Affairs
 Read More
Read More
Copan Launches UriVerse™: Fully Automated Solution for Urine Specimen Aliquoting
 Read More
Read More
Hospital Angeles Pedregal Makes History with Latin America's First Copan WASPLab®
 Read More
Read More
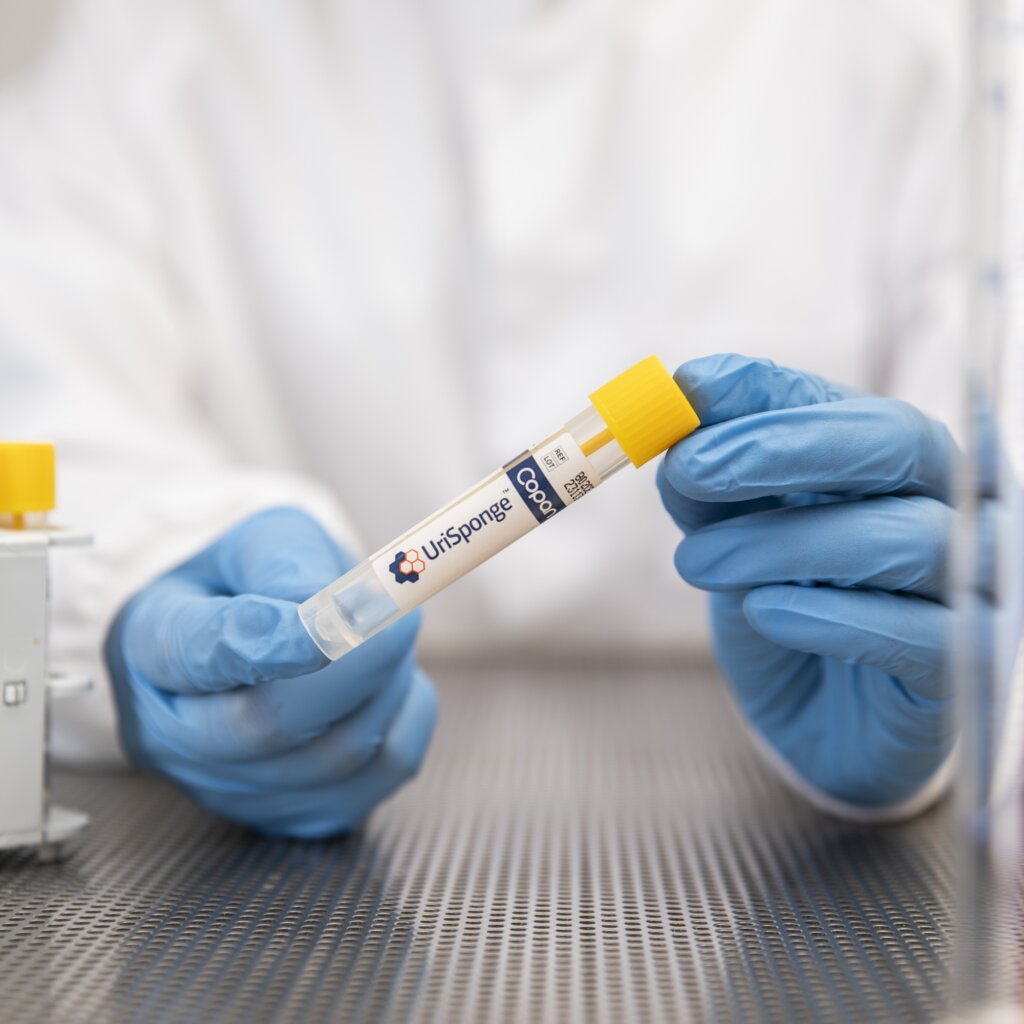
FDA Clears UriSponge®: A Streamlined, Cost-Effective Solution for Urine Specimen Collection and Transport
 Read More
Read More
Latest Company News
Copan Expands U.S. Manufacturing Footprint with Multimillion-Dollar Investment
Copan Diagnostics, part privately held Italy-based Copan Group and a leader in specimen collection and transport, and laboratory automation, announced over $7 million in new U.S. manufacturing investments in California and Puerto Rico. This expansion underscores Copan’s commitment to serving the U.S. market with locally manufactured products.
The investment includes facility expansions, new manufacturing processes, advanced quality control systems, and implementation of new production technologies in the U.S. This initiative accelerates production of critical components and finished goods, such as UTM® Universal Transport Medium kits, used for collecting respiratory viral specimens, and UriSponge®, Copan’s novel urine collection and transport device – reducing reliance on overseas supply chains and enabling faster response to U.S. customer needs.
U.S. Manufacturing Highlights
- Over $5 million to expand production capacity at Copan’s California and Puerto Rico facilities
- $1 million for U.S.-based production of UriSponge®
- $1 million to increase sourcing of components and reagents from U.S. suppliers
- New centralized QA laboratory to strengthen quality control, innovation, and efficiency
“While Copan is proudly rooted in Italy, our growth strategy is deeply connected to the U.S. market,” said Stefania Triva, President of Copan Group. “Expanding our U.S. manufacturing footprint is a key part of our plan to invest closer to critical markets and better serve our customers.”
“With more than three decades of United States presence, we remain committed to fostering economic growth by strengthening local manufacturing, increasing access to advanced technologies, and creating new employment opportunities in our communities,” added Fabrizio Mazzocchi, CEO of COPAN Diagnostics Inc.
Construction began this month, with operations projected to launch in Q1 2026.
Strengthening U.S. Presence
Copan’s U.S. operations include more than 450 employees nationwide. By increasing local production and sourcing, Copan is not only enhancing supply chain resilience but also investing in the future of U.S. laboratories.
Copan’s products and technologies are used daily by laboratory professionals and healthcare providers across the United States.
About Copan Diagnostics, Inc.
Copan Diagnostics is part of Copan Group, a leading global manufacturer of collection and transport systems. Through its collaborative approach, Copan has developed breakthrough technologies such as FLOQSwabs®, ESwab®, UTM® Universal Transport Medium™, and WASPLab®, Full Laboratory Automation with Artificial Intelligence. Copan continues to innovate and transform collection and transport systems, as well as laboratory automation, helping healthcare providers improve patient care.
September 24, 2025

FAQ’s
UriSponge® FAQ
Explore the UriSponge® FAQ page to learn about our advanced, boric acid-free urine specimen preservation and transport system. Discover its uses, safety features, operational benefits, and how it streamlines workflows for modern laboratories.
WASPLab® FAQ
The WASPLab® FAQ is an extensive resource that offers in-depth information about Copan's cutting-edge automated digital microbiology platform, addressing essential aspects such as system features, benefits, technical details, workflow optimization, artificial intelligence integration, customization options, and return on investment.
WASP® FAQ
The WASP® FAQ is a comprehensive guide that provides detailed information about Copan's automated microbiology specimen processing system, covering key topics such as capabilities, advantages, technical specifications, quality control, integration, and cost considerations.
UniVerse® FAQ
This FAQ provides detailed insights into the COPAN UniVerse® system, a cutting-edge automated platform for specimen preparation. It covers key aspects such as functionality, applications, benefits, compatibility, and technical specifications, offering a thorough understanding of how UniVerse® enhances laboratory operations and efficiency.
Colibrí™ FAQ
Explore Copan's Colibrí™ system for automated colony picking and MALDI-TOF/AST sample prep. Learn how Colibrí integrates with WASPLab® and PhenoMATRIX®, improves lab efficiency, and ensures traceability. Get answers to common questions in our detailed Colibrí FAQ.
FLOQSwabs® FAQ
This FAQ page provides information about flocked swabs, including what they are, how they were invented by Copan, why Copans's FLOQSwabs® are superior, the research backing their performance and use for flu and COVID-19 testing.
eNAT® FAQ
This FAQ page provides information on eNAT®, a sample collection and transport system that stabilizes and preserves nucleic acids for molecular testing, covering topics such as what eNAT® is, how it works, applications, compatibility, and availability.
ESwab® FAQ
This FAQ page provides information on ESwab®, a liquid-based multipurpose collection and transport system for microbiology specimens, covering topics such as what ESwab® is, how to use it, what it supports, stability requirements, applications, and availability.
UTM® FAQ
This FAQ page provides information on UTM®, a viral transport medium. Topics covered include what UTM® is, how it is used, what is in the formulation, viability duration, and availability.
PodSwab™ FAQ
This FAQ covers key questions about how PodSwab™ works and the science behind pooled testing. We explain what PodSwab™ is, how sample pooling allows efficient use of tests, directions for use, and how to set up a program.
FecalSwab® FAQ
This FAQ page provides information on FecalSwab®, a collection, transport and processing system for enteric bacteria. It covers topics such as what FecalSwab® is, how to use it, applications, and availability.
Recent Blog Posts
Study Validates Effectiveness of Copan FecalSwab® for Enteric Pathogen Detection on the BD MAX™ System
August 26, 2024

Comprehensive Guide to Cary-Blair Transport Medium: The Gold Standard for Fecal Sample Collection and Transport
August 23, 2024

Study Presented at CACMID 2024 Highlights Copan’s UniVerse® as a Game-Changer in Clinical Molecular Specimen Preparation
July 22, 2024

Research Roundup
Dive into the articles below for research related to COPAN products as well as many other aspects of sample collection and transport, automation, food and pharmaceutical testing, forensic and genetic collection and more!
Research RoundUp: FecalSwab® and Molecular Testing
March 7, 2023

Research Roundup: ASM Microbe 2022 Scientific Posters on Colibrí®
June 28, 2022

Research Roundup: ESwab® Can Do All That?!
February 11, 2022
Upcoming Events
No events message







