New Study Shows the Impacts of the Variability in Instructions for Performance of Nasopharyngeal Swabs in the Era of COVID-19
February 11, 2021
The gold standard collection to determine the presence of SARS-CoV-2 uses a nasopharyngeal (NP) swab. Given the significant demand for COVID19 testing, there are a large number of previously untrained and unfamiliar staff now collecting these specimens. We have all seen a variety of ways that nasopharyngeal swabs are actually being collected from having a specimen collected on ourselves or seeing collections performed on the television or other media.
A group of researchers in Canada noticed that there was significant differences in instructions for performing NP swab collections across the country, so they embarked on comparing these to those provided by the Centers for Disease Control and Prevention (CDC), the Pan American Health Organization (PAHO), and to instructions from the New England Journal of Medicine.

Instructions for NP swab collections were obtained from 81 regional health authorities: 10/13 (77%) of the instructions indicated that the patient should clear their nasal passages before swab insertion, 11/13 (85%) instructed that the patient’s head should be tilted back slightly for collection, 12/13 (92%) instructed the collector to insert the swab parallel to the palate, but only 3/13 (23%) instructed that the swab should be inserted to at least a depth of two-thirds the distance between the patient’s nose and ear. For example, one area provided a pictogram of the depth of swab insertion but no distance or technique for estimating this depth was given, 6 areas recommended that 4cm or one-half the distance from nostrils to ear should be the length of insertion, 2 recommended 7cm of depth for insertion, 1 recommended one-half to two-thirds the distance from nose to ear, and 3 recommended an insertion from the nose to the external ear canal.
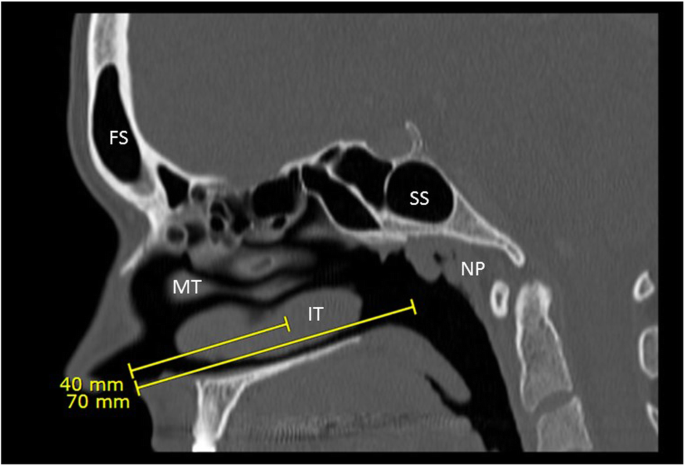
This study concluded that there are significant differences across Canada in guidance for obtaining nasopharyngeal swab specimens. They also determined that the majority of instructions would not be effective in the swab reaching the nasopharynx for an appropriate collection. If not inserted deep enough, a mid-nasal or mid-turbinate swab is performed instead, potentially reducing the sensitivity of the swab.
A recent article and accompanying video in NEJM (New England Journal Medicine 2020; 382:e76) outlines the correct way to obtain a nasopharyngeal swab specimen. See this article for further details.
Illustrated Sampling Guides for COVID-19
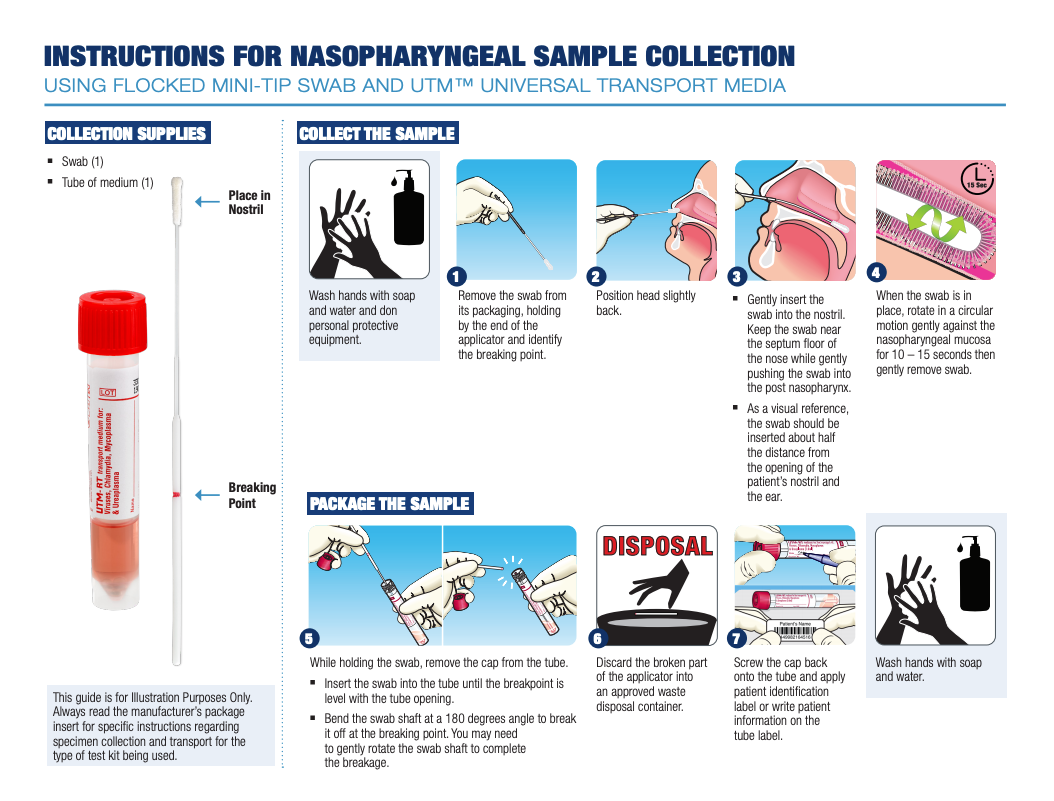
Below are examples of guides that can be used to illustrate how to collect viral specimens from different sites using Copan ESwab™ and UTM®: Universal Transport Medium™ collection kits. Always read the manufacturer’s package insert for specific instructions regarding specimen collection and transport for the type of test kit being used.


Nasopharyngeal Collection |
Oropharyngeal Collection |
Anterior Nares Collection |
| UTM® Collection Guide | UTM® Collection Guide | UTM® Collection Guide |
| ESwab™ Collection Guide | ESwab™ Collection Guide | ESwab™ Collection Guide |

