FecalSwab®, An Excellent Alternative When Utilizing BD MAX™ Enteric Bacterial Panel and the Extended Enteric Bacterial Panel, Study Finds
April 22, 2020
Gastrointestinal (GI) diseases are common illnesses, affecting more than 1.6 million people annually and account for a significant portion of medical burden and expenditure worldwide. GI diseases are so costly, that it is estimated the annual healthcare expenses in the United States are greater than many prevalent diseases, such as heart disease, trauma, or mental health conditions. Diarrheal diseases can have numerous causes and proper diagnosis can be challenging due to the diversity of organisms and potential pathogens found in stool.
Conventional microbiology is the most common method of detecting and diagnosing GI diseases, but it has some drawbacks. Traditional microbiology methods are labor-intensive, time-consuming, and often require days of incubation prior to analysis by highly-skilled laboratory personnel. For these reasons, molecular platforms are beginning to gain traction in the clinical microbiology market. One of these platforms, developed by Becton Dickinson, is the BD MAX™ system, a molecular analyzer that can test up to 24 samples at once in approximately 3 hours with minimal intervention from laboratory staff. The system recommends using the bulk stool specimen transport device Meridian Para-Pak® with Cary-Blair medium. However, obtaining sufficient samples to fill the bulk stool collection systems could be challenging from certain patients, potentially causing delays.
A new study published in Diagnostic Microbiology & Infectious Disease examines the use of low volume COPAN FecalSwab® specimens on the BD MAX™ molecular platform. The objective of the study is to determine the ideal volume of the flocked swab-based FecalSwab® medium equivalent to the BD MAX™ recommended bulk stool device Meridian Para-Pak Cary-Blair medium. Not only did FecalSwab® perform as well as Cary-Blair bulk stool specimens, but the study suggested COPAN FecalSwab® has many practical advantages.
In this study, 144 unpreserved stool specimens from Tampa General Hospital were spiked with known strains of bacteria that commonly cause GI diseases. These specimens were then used to inoculate both the COPAN FecalSwab® and bulk stool Para-Pak® device and tested in triplicate.
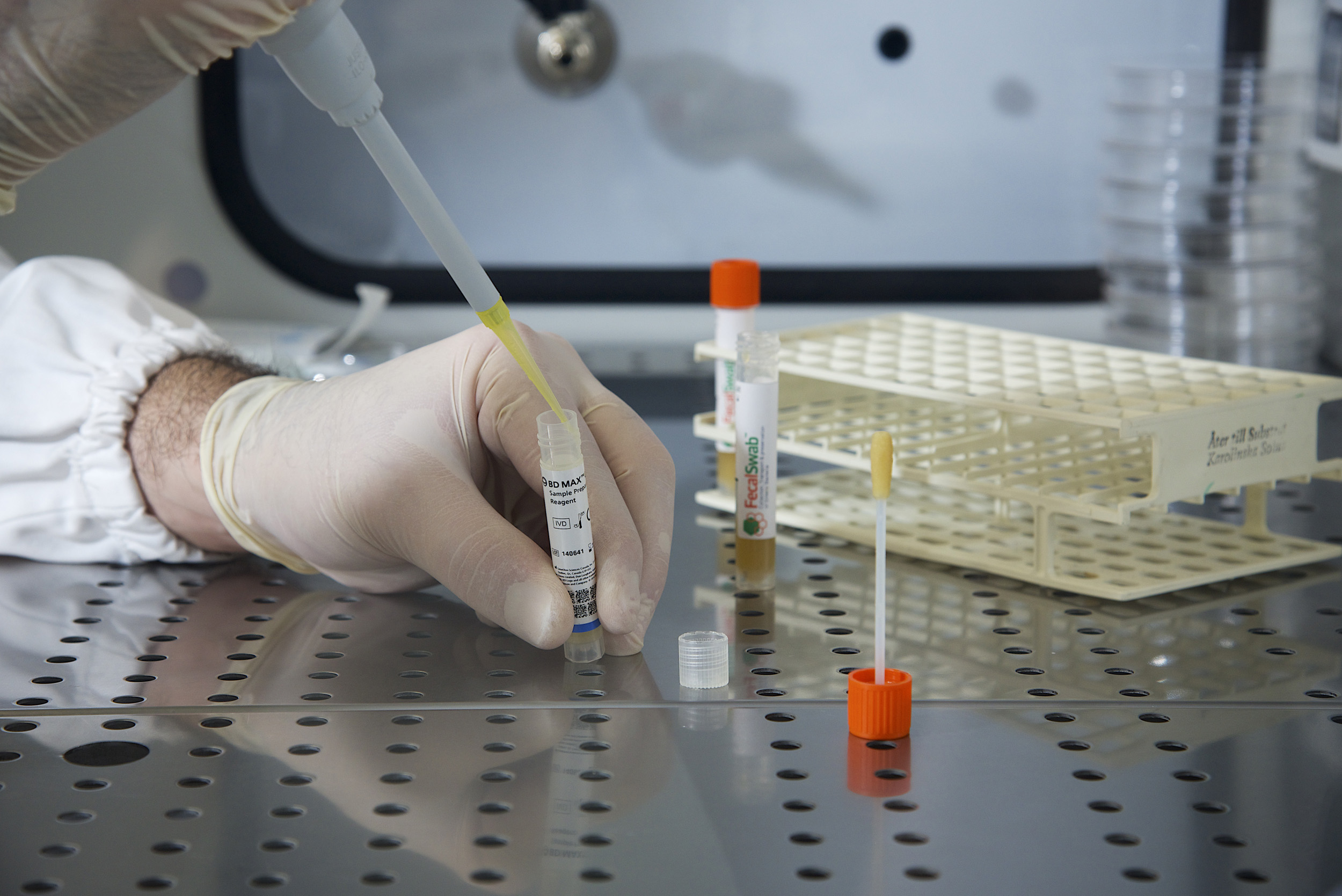
The study found a 100% agreement between FecalSwab® and the BD MAX™ recommended Para-Pak® medium. Further, the study found that the performance of 50 μl of FecalSwab® preserved samples was equivalent to 10 μl of traditional Cary-Blair Para-Pak® preserved specimens for GI pathogen detection using the BDM GIP. It was not surprising to the researchers that FecalSwab® requires a higher volume than Para-Pak®, because FecalSwab® collects a much smaller stool specimen.
The ability of the FecalSwab® stool transport medium to preserve the lower volume specimens and maintain the integrity of nucleic acids at different storage conditions was also examined. The data shows that the test performance is not altered when FecalSwab® specimens are stored for at least 24 hours at room temperature or 5 days at 2-8°C. Not only does FecalSwab® performs equivalent to Para-Pak® under a variety of conditions, but using FecalSwab® has been found to have several distinct practical advantages.
FecalSwab® combines a COPAN-invented flocked swab with 2mL of Cary-Blair medium in a plastic, screw cap tube. The flocked swab tip is coated with perpendicularly sprayed-on Nylon® fibers. This coating creates a thin absorbent layer that allows for quick sample uptake and elution of more than 90% of the sample. The improved absorption of bacteria, as well as a greater transference of material onto Gram stain slides, can help improve detection rates and increase the sensitivity of diagnostic tests.
Further, collection and transport devices, like Para-Pak®, require bulk stool samples that are often difficult to collect because some patients may be unable or too dehydrated from GI symptoms to produce a sufficient sample. It can be especially difficult to collect sufficient stool specimens from children and infants. FecalSwab® requires less sample volume that can be taken directly from the patient at the time of the visit with a healthcare provider without delay. This shorter time to the collection using the FecalSwab™ can provide a faster result turnaround time than bulk stool specimens, indicating that the use of the flocked swab system may lead to improvements within the clinical workflow, as well as potential outcomes. These advantages along with the increasing body of evidence supporting the use of FecalSwab® on molecular platforms, like BD MAX™, make it easy to see why COPAN FecalSwab® is used by more and more laboratories and medical institutions around the world. Sometimes, less is more!
Reference
H.F. Rojas, A. Lima, C. Kubasek, et al., Evaluation of Copan FecalSwab™ Preserved Stool Specimens with the BD MAX™ Enteric Bacterial Panel and the BD MAX™ Extended Enteric Bacterial Panel, Diagnostic Microbiology & Infectious Disease (2020), https://doi.org/10.1016/j.diagmicrobio.2020.115055

